Abstract
[Background] Minor populations (0.003%-1.0%) of glycosylphosphatidylinositol-anchored protein-deficient granulocytes (GPI[-] Gs) are often detected in the peripheral blood (PB) of patients with acquired aplastic anemia (AA) and low-risk myelodysplastic syndromes and are thought to represent immune pathophysiology of bone marrow failure. We previously reported that minor GPI(-) G populations were detected in some healthy individuals (HIs) and persisted over several years at similar percentages (Katagiri T, et al. Stem Cells 2013). Several lines of evidence have suggested that small numbers of GPI(-) Gs detected in HIs are polyclonal populations mostly derived from short-lived PIGA-mutated committed progenitor cells. Minor GPI(-) G populations in AA patients may also be derived from multiple committed progenitor cells rather than from a few hematopoietic stem progenitor cells (HSPCs) with PIGA mutations. However, minor GPI(-) G populations usually persist for a long period of time at similar frequencies in AA patients, suggesting that they may instead be derived from a few HSPCs that have undergone PIGA mutations. This issue remains debated due to the inability to sequence the PIGA gene in the very few GPI(-) granulocytes available. We recently developed a sensitive method capable of detecting PIGA mutations in minor GPI(-) Gs using amplicon sequencing of GPI(-) Gs that were enriched with magnetic microbeads followed by FACS sorting. Using this method, we addressed whether minor GPI(-) G populations in AA patients and HIs are oligoclonal or polyclonal as well as which cell population they are derived from HSPCs or committed progenitor cells.
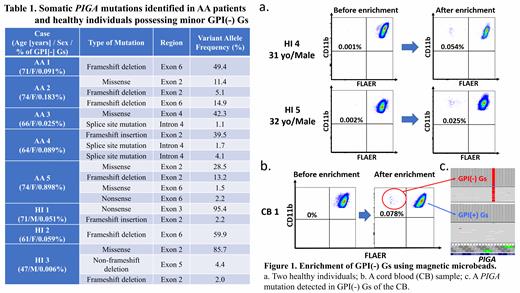
[Methods] Five AA patients possessing 0.025%-0.898% GPI(-) Gs, 3 HIs who were found to have ≥0.003% (0.006%, 0.051%, and 0.059%) GPI(-) Gs during a screening of more than 200 HIs for GPI(-) Gs, 30 HIs (median: 37 years old, male/female:17/13) with 0% to 0.002% GPI(-) Gs, and 8 cord blood (CB) samples were subjected to enrichment of GPI(-) Gs for PIGA sequencing. Their leukocytes were treated with PE-labelled anti-CD55 monoclonal antibodies (mAbs) and anti-CD59 mAbs, and CD55 +CD59 + granulocytes were removed using magnetic microbeads labelled with anti-PE mAbs. CD11b +FLAER-negative granulocytes were sorted from the remaining granulocytes using FACSAria Fusion. DNA from sorted GPI(-) Gs was amplified using primers covering all exons of PIGA. Nucleotide sequences of the PIGA gene in GPI(-) Gs were determined using a next-generation sequencer.
[Results] This novel enrichment method enabled the detection of only 1-4 different PIGA mutations in all 5 female AA patients (AA 1-5) with the total of different VAFs in each case reaching nearly 50% (Table 1). Limited kinds of PIGA mutations were also detected in three HIs (two males [HI 1 and 3] and one female [HI 2]). For HI 1 and HI 3, the VAFs of predominant PIGA-mutated sequences were longitudinally measurable using whole-blood DNA samples with droplet digital PCR, which showed no apparent changes in the VAF (0.020%-0.027% for HI 1 and 0.012%-0.025% for HI 3 over 4 and 6 years, respectively). The presence of mono or oligoclonal GPI(-) Gs in the 3 HIs prompted us to study 30 HIs who had been judged to be negative for minor GPI(-) G populations by a high-sensitivity flow cytometry method. The enrichment method unexpectedly identified clear CD11b highFLAER - GPI(-) G clusters in granulocytes from 24 of the 30 HIs (Figure 1a). The median number of GPI(-) Gs contained in 7 ml of PB was 31 (range, 1-136 cells). Sufficient amounts of DNA for NGS were obtained from sorted GPI(-) Gs of six subjects, and PIGA amplicon sequencing revealed 1-3 different PIGA mutations in four of the six subjects. The examination of fresh CB also revealed clear GPI(-) G clusters in four of eight samples (Figure 1b). PIGA amplicon sequencing of 79 GPI(-) Gs obtained from 1 male CB sample (CB 1) showed a sole PIGA mutation with VAFs of 95% (Figure 1c).
[Conclusion] Minor GPI(-) G populations detectable in patients with AA and HIs are derived from a few PIGA-mutated HSPCs, not from committed myeloid progenitor cells, a finding that negates a hypothesis that a few PIGA-mutated HSPCs are selected from polyclonal PIGA-mutated HSPCs during transition from AA to florid PNH. Very small numbers of GPI(-) Gs are present much more frequently in HIs than previously thought and may also be derived from a few HSPCs with PIGA mutations that occur in HSPCs during the fetal stage.
Takamatsu: Bristol-Myers Squibb: Honoraria, Research Funding; Adaptive Biotechnologies, Eisai: Honoraria; SRL: Consultancy; Janssen: Consultancy, Honoraria, Research Funding. Yamazaki: Novartis Pharma: Honoraria; Kyowa Kirin: Research Funding; Kyowa Kirin: Honoraria. Nakao: Symbio: Consultancy; Kyowa Kirin: Honoraria; Novartis Pharma: Honoraria; Alexion Pharma: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal